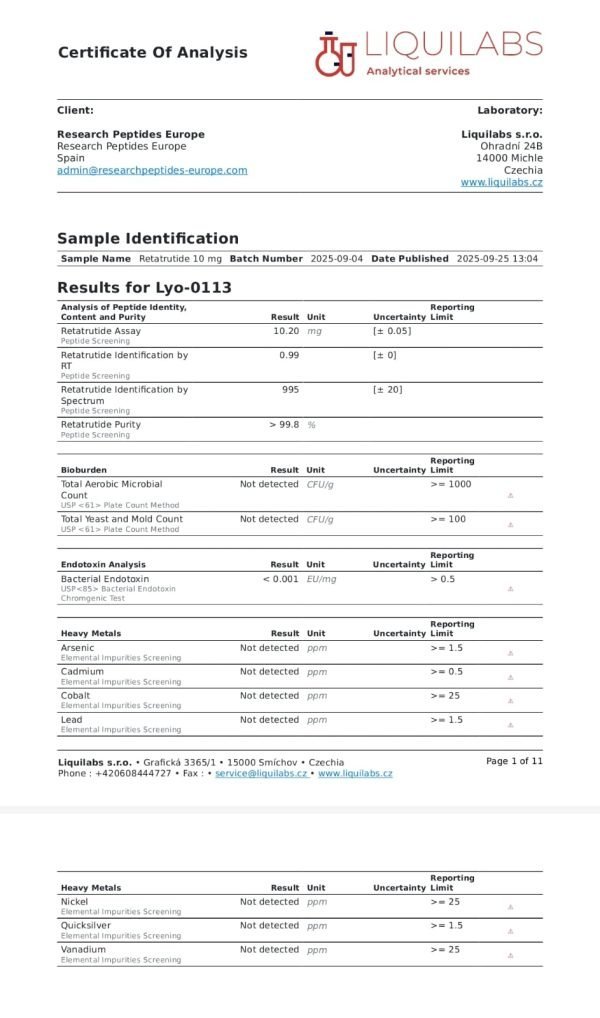
Retatrutide 20mg
€229.99
Technical Specifications
| Specification | Detail |
|---|---|
| Product Name | Retatrutide (LY3437943) – R&D Only |
| Quantity | 20mg per vial |
| Purity | ≥99% (HPLC verified) |
| Form | Lyophilized powder |
| Appearance | White to off-white lyophilized powder |
| Molecular Weight | 4731.33 g/mol |
| CAS Number | 2381089-83-2 |
| Amino Acid Sequence | Tyr-{Aib}-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Ile-{α-Me-Leu}-Leu-Asp-Lys-{diacid-C₂₀-γGlu-(AEEA)-Lys}-Ala-Gln-{Aib}-Ala-Phe-Ile-Glu-Tyr-Leu-Leu-Glu-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro-Pro-Ser-NH₂ |
| Peptide Length | 39 amino acids |
| Target Receptors | GLP-1R, GIPR, GCGR |
| Mechanism | Triple hormone receptor agonist |
| Storage (Lyophilized) | -20°C; protect from light and moisture |
| Shipping | Ambient temperature with protective packaging |
| Intended Use | Research and laboratory use only |
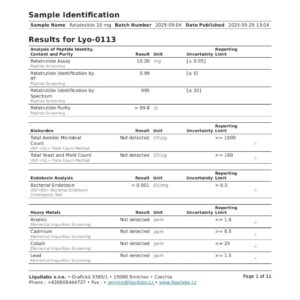
| Documentation | Certificate of Analysis provided with each batch |
Premium Retatrutide (LY3437943) for Sale | 20mg Lyophilized Powder | ≥99% Purity | 48-Hour Delivery Across EU & UK
Retatrutide (LY3437943): The Revolutionary Triple Agonist Redefining Metabolic Research
eupeptidelap.co.uk is proud to present Retatrutide 20mg, a premium research-grade triple agonist peptide manufactured to the highest analytical standards. As a trusted peptide vendor uk and leading EU peptide supplier, we provide researchers across Europe with Retatrutide for sale that delivers exceptional purity, consistency, and documented quality for advanced studies in metabolic regulation, obesity, and endocrine research .
Retatrutide (LY3437943) represents a groundbreaking advancement in metabolic research as the first-in-class triple agonist peptide targeting three key hormone receptors simultaneously: the glucagon-like peptide-1 (GLP-1) receptor, the glucose-dependent insulinotropic polypeptide (GIP) receptor, and the glucagon (GCG) receptor . This sophisticated 39-amino acid synthetic peptide is engineered to harness the complementary actions of all three incretin hormones, producing unprecedented metabolic effects that surpass even the most advanced dual agonists currently available .
The molecular formula of Retatrutide is C₂₂₁H₃₄₂N₄₆O₆₈, with a molecular weight of 4731.33 g/mol . Its unique structural design includes three non-coding amino acid residues at positions 2, 13, and 20—Aib2 (α-aminoisobutyric acid) provides resistance to DPP-4 degradation, Aib20 contributes to optimal GIP activity, and αMeL13 enhances GCG and GIP receptor activation . The peptide backbone is conjugated to a C20 fatty diacid moiety via a linker at the lysine residue at position 17, enabling albumin binding as a pharmacokinetic half-life extension strategy .
The triple agonism approach leverages:
-
GLP-1 Receptor Activation: Enhances insulin secretion in response to glucose, slows gastric emptying, and acts directly on brain appetite centers to suppress hunger
-
GIP Receptor Activation: Stimulates insulin release, influences fat metabolism and storage, and may affect brain reward pathways associated with food
-
Glucagon Receptor Activation: Increases energy expenditure, promotes lipolysis (fat breakdown), and reduces fat accumulation in the liver—while the potent glucose-lowering effects of GLP-1 and GIP offset glucagon’s potential blood sugar-raising effects
For researchers seeking to buy peptide online EU for investigations into metabolic disorders, obesity pharmacotherapy, type 2 diabetes, or complications of obesity such as osteoarthritis, eupeptidelap.co.uk offers this premium research compound with comprehensive documentation, including Certificates of Analysis and batch-specific purity data . Whether your laboratory is based in London, Berlin, Paris, or anywhere in the European Union, our guaranteed 48 hour delivery peptide service ensures your research continues without interruption.
The Scientific Foundation of Retatrutide Research
The Evolution from Single to Triple Agonism
The development of retatrutide represents the culmination of decades of research into incretin hormones and their role in metabolic regulation . First-generation therapies (GLP-1 receptor agonists like semaglutide) targeted a single hormone pathway, achieving approximately 15% average weight loss at 68 weeks . Second-generation dual agonists (GIP/GLP-1 agonists like tirzepatide) improved outcomes to approximately 21-22% weight loss .
Retatrutide, as the first triple agonist, has demonstrated the most robust results yet, with Phase 2 trials showing 24.2% weight loss at 48 weeks—one of the highest weight loss percentages of any pharmacological therapy . This progressive improvement illustrates the power of multi-receptor targeting in metabolic research.
Mechanism of Action: Triple Receptor Activation
Retatrutide exerts its effects through simultaneous activation of three key receptors, creating synergistic actions that exceed what any single pathway could achieve alone .
GLP-1 Receptor Activation: Glucagon-like peptide-1 is an incretin hormone that helps regulate appetite and signals satiety. It stimulates insulin release, lowering blood sugar, and acts on the central nervous system to reduce appetite while slowing gastric emptying .
GIP Receptor Activation: Glucose-dependent insulinotropic polypeptide is another incretin hormone released during eating. Like GLP-1, it acts as a satiety signal and increases insulin production, while also helping regulate fat metabolism and blood sugar. Activating GIP receptors is thought to enhance the effects of GLP-1 when these two actions are combined .
Glucagon Receptor Activation: The addition of glucagon agonism is what truly distinguishes retatrutide from other metabolic peptides . While glucagon alone would raise blood sugar—potentially counteracting weight loss efforts—when activated alongside GLP-1 and GIP receptors, it increases overall energy expenditure and fat burning without excessively raising blood glucose . This novel combined mechanism means retatrutide is the first metabolic peptide to target three pathways, effectively adding an “efficient combustion engine” to the “fuel limiter” effects of GLP-1 and GIP .
In Vitro Activity and Receptor Selectivity
Retatrutide has been carefully engineered to achieve balanced activation of all three receptor types. In vitro studies using HEK-293 cell lines expressing human receptors demonstrate :
-
GIP Receptor Activity: Retatrutide exhibits potent GIP receptor agonism with an EC50 of 0.0643 nM, demonstrating approximately 8.9-fold greater potency than native human GIP(1-42)-NH₂
-
GLP-1 Receptor Activity: The peptide activates human GLP-1 receptors with an EC50 of 0.775 nM, approximately 2.5-fold less potent than native GLP-1(7-36)-NH₂
-
Glucagon Receptor Activity: Retatrutide activates human GCG receptors with an EC50 of 5.79 nM, approximately 2.9-fold less potent than native human glucagon
This balanced activation profile ensures that all three pathways contribute meaningfully to the compound’s metabolic effects, with the high GIP potency potentially driving enhanced adipocyte engagement and lipid metabolism .
Key Research Applications
Obesity and Weight Loss Research
Retatrutide is extensively used in studies investigating body weight regulation, adipose tissue biology, and energy homeostasis. Clinical trials have demonstrated unprecedented weight loss results that set a new high bar for obesity pharmacotherapy research .
Phase 2 Clinical Trial Results: The landmark Phase 2 randomized, double-blind, placebo-controlled trial published in the New England Journal of Medicine (2023) enrolled 338 adults with BMI ≥30 (or ≥27 with a weight-related comorbidity) without type 2 diabetes. Results after 48 weeks showed :
-
Unprecedented Weight Loss: Participants receiving 12 mg weekly achieved a mean weight reduction of 24.2% of initial body weight—translating to approximately 26.4 kg (58 pounds)
-
Dose-Dependent Efficacy: Weight loss effects were clearly dose-dependent
-
Continued Weight Loss Trajectory: Notably, the weight loss trajectory for the highest dose groups had not yet plateaued at the end of the 48-week study, suggesting that longer treatment duration could lead to even greater weight reduction
-
Normalization of Prediabetes: Every participant with prediabetes at baseline in the 8 mg and 12 mg groups reverted to normal glycemic status by study end
Mechanism Differentiation: Unlike dual agonists that primarily reduce weight through caloric restriction, retatrutide’s addition of glucagon agonism increases energy expenditure, creating a dual-mechanism approach to weight reduction .
Type 2 Diabetes Research
Retatrutide has demonstrated exceptional efficacy in glycemic control research :
-
Phase 2 Diabetes Study: In 281 T2D subjects, retatrutide improved glycemic control more than dulaglutide, with HbA1c reductions as great as 2.16% with retatrutide versus 1.36% with dulaglutide at 36 weeks
-
Superior Efficacy: The triple agonist approach produced greater improvements in glycemic control than selective GLP-1 receptor agonism alone
-
Mechanism: The glucose-lowering effects of GLP-1 and GIP receptor activation offset any potential hyperglycemic effects of glucagon agonism, creating a self-regulating system
Non-Alcoholic Fatty Liver Disease (NAFLD/NASH) Research
A particularly exciting finding from Phase 2 trials was retatrutide’s powerful effect on liver fat :
-
Baseline Liver Fat: Participants had average liver fat content of approximately 20%
-
Post-Treatment Reduction: After 48 weeks on 12 mg dose, liver fat decreased by an average of over 86%
-
Complete Resolution: Remarkably, nearly 86% of participants in the highest dose group achieved complete resolution of excess liver fat (below 5%)
-
Mechanism: Glucagon receptor agonism directly promotes hepatic fat oxidation and reduces de novo lipogenesis, making retatrutide uniquely positioned for NAFLD/NASH research
Cardiometabolic Research
Beyond weight loss, participants saw significant improvements in key health markers :
-
Blood Pressure Reduction: Dose-dependent decreases in both systolic and diastolic blood pressure
-
Lipid Profile Improvement: Significant, dose-dependent reductions in triglycerides, along with increases in beneficial HDL cholesterol
-
Glycemic Normalization: As noted above, complete normalization of prediabetes in all participants on the two highest doses
Obstructive Sleep Apnea Research
The TRIUMPH clinical development program includes dedicated evaluation of retatrutide for obstructive sleep apnea (OSA), a common complication of obesity . The Phase 3 TRIUMPH-1 and TRIUMPH-2 trials include OSA-specific endpoints, measuring change in Apnea-Hypopnea Index (AHI) as a primary outcome . Given that substantial weight reduction is often required to meaningfully improve OSA, retatrutide’s unprecedented efficacy positions it as a particularly promising candidate for OSA research.
Knee Osteoarthritis Research
The TRIUMPH-4 trial is specifically designed to evaluate retatrutide in adults with obesity or overweight and knee osteoarthritis . Key endpoints include change in the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain subscale score, reflecting the growing recognition that substantial weight loss can significantly improve joint pain and function .
Ongoing Phase 3 TRIUMPH Program Research
Based on highly successful Phase 2 results, Eli Lilly has launched an extensive Phase 3 clinical trial program (TRIUMPH) evaluating retatrutide in diverse populations and for different indications, enrolling over 5,800 participants :
| Trial | Population | Primary Focus |
|---|---|---|
| TRIUMPH-1 | Weight management | Obesity with OSA and/or OA protocols nested within |
| TRIUMPH-2 | Weight management | Obesity with type 2 diabetes and OSA and/or OA protocols nested within |
| TRIUMPH-3 | Weight management | Population with cardiovascular disease |
| TRIUMPH-4 | Stand-alone OA trial | Knee osteoarthritis |
Quality Assurance: Setting the Standard for Research Compounds
Manufacturing Excellence
eupeptidelap.co.uk sources Retatrutide 20mg from certified GMP facilities with rigorous quality control protocols :
-
HPLC Purity Analysis: ≥99% purity verified by high-performance liquid chromatography
-
Mass Spectrometry Verification: Molecular weight confirmation (4731.33 g/mol) via LC-HRMS
-
Batch-Specific Certificates of Analysis: Complete documentation for each production run
-
Research Use Only (RUO) Labeling: Full compliance with research compound regulations
Chemical and Product Specifications
| Specification | Detail |
|---|---|
| Product Name | Retatrutide (LY3437943) – R&D Only |
| CAS Number | 2381089-83-2 |
| Molecular Formula | C₂₂₁H₃₄₂N₄₆O₆₈ |
| Molecular Weight | 4731.33 g/mol |
| Amino Acid Length | 39 amino acids |
| Purity | ≥99% (HPLC verified) |
| Appearance | White to off-white lyophilized powder |
| Form | Lyophilized powder |
| Solubility | Soluble in aqueous solution adjusted to pH ~9 with NH₃·H₂O |
| Mechanism | Triple GLP-1/GIP/Glucagon receptor agonist |
| Storage (Lyophilized) | -20°C; protect from light and moisture |
| Storage (Reconstituted) | 2-8°C for short-term; aliquot and store at -80°C for long-term |
| Half-Life | Approximately 134-165 hours |
| Shipping | Ambient temperature with protective packaging |
| Intended Use | Research and laboratory use only |
| Documentation | Certificate of Analysis provided with each batch |
Stability and Handling Guidelines
Lyophilized Powder Storage: Store lyophilized retatrutide powder at -20°C in a dry, dark environment, protected from light and moisture . Under proper storage conditions, the powder maintains stability for extended periods.
Reconstitution Protocol :
-
Allow vial and solvent to reach ambient temperature before opening to prevent condensation
-
Reconstitute using appropriate solvent according to research protocol (some protocols recommend 50% bacteriostatic water and 50% acetic acid for optimal results)
-
Inject solvent slowly down the inside wall of the vial to minimize foaming
-
Gently swirl or roll the vial until fully dissolved—do not shake vigorously
-
Label the vial with the date of reconstitution
-
Refrigerate immediately at 2-8°C
Reconstituted Solution Storage: Store reconstituted solutions at 2-8°C for short-term use. For longer-term storage, aliquot into single-use portions and store at -80°C. Avoid repeated freeze-thaw cycles as this can degrade the peptide.
Protection from Light and Moisture:
-
Store in original packaging protected from light
-
Keep vials tightly sealed when not in use
-
Allow refrigerated vials to reach room temperature before opening to prevent condensation
Important Note: Retatrutide is soluble and stable in aqueous solution adjusted to pH ~9 with NH₃·H₂O . Always follow established laboratory protocols for peptide handling.
Key Benefits
-
Premium Quality Manufacturing: GMP-certified production, ≥99% HPLC-verified purity
-
First-In-Class Triple Agonist: Simultaneous GLP-1, GIP, and glucagon receptor activation for synergistic research applications
-
High Concentration: 20mg per vial—ideal quantity for targeted metabolic studies
-
Unprecedented Weight Loss Research: Enables investigation of 24.2% body weight reduction in obesity models at 48 weeks
-
NAFLD/NASH Research: Reduces liver fat by >86% with nearly complete resolution in Phase 2
-
Glycemic Control Studies: HbA1c reductions up to 2.16% in T2D research models
-
Energy Expenditure Research: Glucagon agonism increases basal metabolic rate through enhanced energy expenditure
-
Cardiometabolic Research: Improves blood pressure, lipid profiles, and insulin sensitivity
-
Appetite Regulation Studies: Triple mechanism targeting hypothalamic and reward pathways
-
Osteoarthritis Research: Dedicated Phase 3 trial evaluating pain and function outcomes
-
Sleep Apnea Research: Being studied in OSA populations with specific AHI endpoints
-
Prediabetes Studies: 100% normalization of prediabetes at higher doses
-
Superior Efficacy: Outperforms dual agonists in head-to-head preclinical comparisons
-
Extended Half-Life: C20 diacid confers albumin binding and prolonged duration (134-165 hours)
-
DPP-4 Resistant: Aib2 modification provides enzymatic stability
-
Comprehensive Documentation: Certificates of Analysis with batch-specific purity data
-
48-Hour EU & UK Delivery: Rapid shipping to research facilities across Europe
-
Batch Consistency: Rigorous quality control ensures lot-to-lot reproducibility
-
EU Sourced: Available from within the European Union
Frequently Asked Questions
Q: Is Retatrutide 20mg suitable for human consumption?
A: No. Retatrutide 20mg from eupeptidelap.co.uk is strictly for research and laboratory use only . It is not for human or animal consumption, and must not be used for therapeutic, diagnostic, or clinical applications. While retatrutide is currently in Phase 3 clinical trials and is expected to reach the market as early as 2026, research-grade material is intended for investigational purposes only . Regulatory agencies have issued warnings against unapproved versions sold without proper RUO status . Researchers must handle this compound in accordance with institutional safety guidelines and local regulations.
Q: Is it legal to purchase retatrutide for research in the UK?
A: Retatrutide is not approved for human use by the MHRA in the UK . The sale of unlicensed peptides under “research use only” labeling is permissible for legitimate research purposes, but products must include explicit RUO labeling and clear disclaimers that they are not for human consumption . At eupeptidelap.co.uk, all products are intended for research and laboratory use only . Legitimate buyers include academic labs, CROs, pharmaceutical R&D, and licensed clinics conducting non-human research. The Medicines and Healthcare products Regulatory Agency (MHRA) may take enforcement action if marketing materials or user reviews indicate personal human use .
Q: What purity level can I expect when I buy retatrutide from eupeptidelap.co.uk?
A: All Retatrutide 20mg from eupeptidelap.co.uk is tested to ≥99% purity by HPLC . Each batch is individually analyzed, and Certificates of Analysis are provided with every order, ensuring you receive material suitable for rigorous research applications.
Q: How effective is retatrutide for weight loss in research models?
A: In clinical trials, retatrutide has demonstrated unprecedented weight loss results. Phase 2 studies at 48 weeks showed 24.2% average weight loss at 12 mg dose—one of the highest weight loss percentages recorded for any pharmacological therapy . The mechanism differs from dual agonists by adding increased energy expenditure to reduced caloric intake .
Q: How should I store retatrutide for long-term stability?
A: Store lyophilized retatrutide powder at -20°C, protected from light and moisture . After reconstitution, store at 2-8°C for short-term use. For longer-term storage, aliquot into single-use portions and store at -80°C. Avoid repeated freeze-thaw cycles.
Q: What is the molecular weight and formula of retatrutide?
A: Retatrutide has the molecular formula C₂₂₁H₃₄₂N₄₆O₆₈ and a molecular weight of 4731.33 g/mol . It is a 39-amino acid peptide containing three non-coding amino acid residues at positions 2, 13, and 20, engineered for balanced activation of GLP-1, GIP, and glucagon receptors .
Q: What is the mechanism of action of retatrutide?
A: Retatrutide is a first-in-class triple receptor agonist targeting GLP-1, GIP, and glucagon receptors simultaneously . GLP-1 and GIP enhance insulin secretion, suppress appetite, and slow gastric emptying, while glucagon increases energy expenditure and promotes fat breakdown . The glucose-lowering effects of GLP-1 and GIP offset glucagon’s potential blood sugar-raising effects, creating a synergistic system .
Q: What research areas commonly use retatrutide?
A: Retatrutide is widely used in obesity and weight loss research, type 2 diabetes research, NAFLD/NASH research, cardiometabolic studies, appetite regulation research, energy expenditure studies, osteoarthritis research, and sleep apnea research .
Q: Do you ship retatrutide to EU countries?
A: Yes. As a dedicated EU peptide supplier, we ship Retatrutide 20mg to all European Union member states with our guaranteed 48 hour delivery peptide service. Our EU fulfilment centre ensures rapid delivery without customs delays. All shipments use protective packaging to maintain compound integrity during transit.
Q: What documentation do you provide with retatrutide orders?
A: Every order includes a Certificate of Analysis (COA) with batch-specific purity data . Additional documentation, including HPLC chromatograms and mass spectrometry data, is available upon request for researchers requiring comprehensive analytical verification. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Q: What are the ongoing clinical trials for retatrutide?
A: The Phase 3 TRIUMPH program includes multiple trials: TRIUMPH-1 (weight management with OSA/OA protocols), TRIUMPH-2 (weight management with T2D and OSA/OA protocols), TRIUMPH-3 (weight management in CVD population), and TRIUMPH-4 (knee osteoarthritis) . These studies have enrolled over 5,800 participants.
Q: Does retatrutide affect liver fat?
A: Yes, dramatically. In Phase 2 studies, participants on 12 mg dose saw liver fat decrease by over 86% from baseline (approximately 20% down to <5%). Nearly 86% achieved complete resolution of excess liver fat . This effect is attributed to glucagon receptor agonism, which directly promotes hepatic fat oxidation and reduces de novo lipogenesis.
Q: What are the common adverse effects observed in retatrutide trials?
A: The most common adverse events are gastrointestinal in nature and dose-related: nausea, diarrhea, vomiting, and constipation . These side effects were most common during the initial dose-escalation phase and tended to be mild to moderate in severity . The safety profile is similar to other incretin mimetics. ...........................................
Q: Do you offer bulk quantities of retatrutide for institutional research?
A: Yes. We accommodate bulk orders for research institutions. Contact our team at sales@eupeptidelap.co.uk for volume pricing, custom requirements, and supply agreements for ongoing research programs.
Advance Your Research with Retatrutide 20mg
eupeptidelap.co.uk is your trusted source for Retatrutide 20mg, the premium choice for researchers investigating metabolic disorders, obesity pharmacotherapy, diabetes, and obesity-related complications. As the first-in-class triple GLP-1/GIP/glucagon receptor agonist, retatrutide represents a revolutionary tool for exploring the synergistic actions of all three incretin pathways and their unprecedented therapeutic potential .
Whether you are exploring weight loss mechanisms, designing diabetes studies, investigating hepatic steatosis, or researching cardiometabolic outcomes, our rigorously tested compound provides the quality and consistency your work demands.
Order today and experience the eupeptidelap.co.uk difference – premium quality, rapid 48-hour delivery across the EU and UK, and expert support for the European research community.
Related products
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU